Summary
Probiotic supplements undergo significant water loss before consumption, killing many of their bacteria and rendering them less effective. Vanderbilt researchers have discovered a protein that protects against damage caused by desiccation and shown that this molecular shield can be added to probiotics to help them survive and colonize the gut. This platform technology can be broadly incorporated into new or existing supplements to make them more efficacious and even improve costs and distribution.
Addressed Need
Substantial amounts of water are removed during the production and distribution of probiotics, especially when they are packaged as solid capsules for convenient storage and consumption. This desiccation causes cellular damage like protein unfolding and aggregation, which can be fatal to the bacteria or reduce their ability to colonize the gut.
Technology Description
This unique protein was identified in a bacteria with an unusual ability to survive desiccation. When expressed heterologously, this protein can protect the cellular machinery of other bacteria from water loss as well, improving their ability to grow again when rehydrated. This protein can also be isolated and mixed with purified enzymes or other therapeutics to preserve them during shipping and storage.
Competitive Advantages
This technology is a platform that could be broadly integrated into new or existing probiotic products. By protecting these probiotics from desiccation, this technology enables the delivery of a higher dosage of viable bacteria, yielding better colonization and thus more efficacious products. Furthermore, this technology reduces the need for other preservation methods like stabilizing agents and refrigeration, thereby driving down costs of production, transportation, and storage and allowing broader distribution.
Stage of Development and IP Status
This technology has been validated in vitro and in vivo. We are seeking commercial partners to further develop it for clinical applications.
Patent: WO2022020397
Publication: Cell Host & Microbe 2022
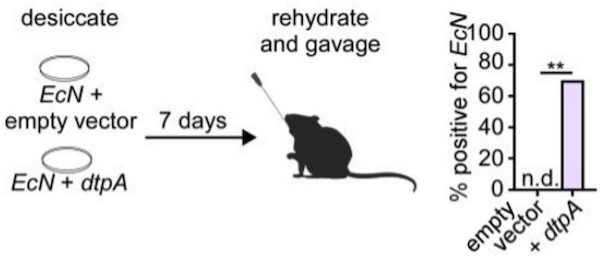
Figure: Cultures of the EcN probiotic strain were engineered to con-tain either the gene encoding the desiccation tolerance protein (dtpA) or an empty vector. These cultures were desic-cated for 7 days, rehydrated, and delivered to mice by oral gavage. While no bacteria could be detected in mice that were administered the control, the bacteria containing the protein successfully colonized the gut of most test mice.




