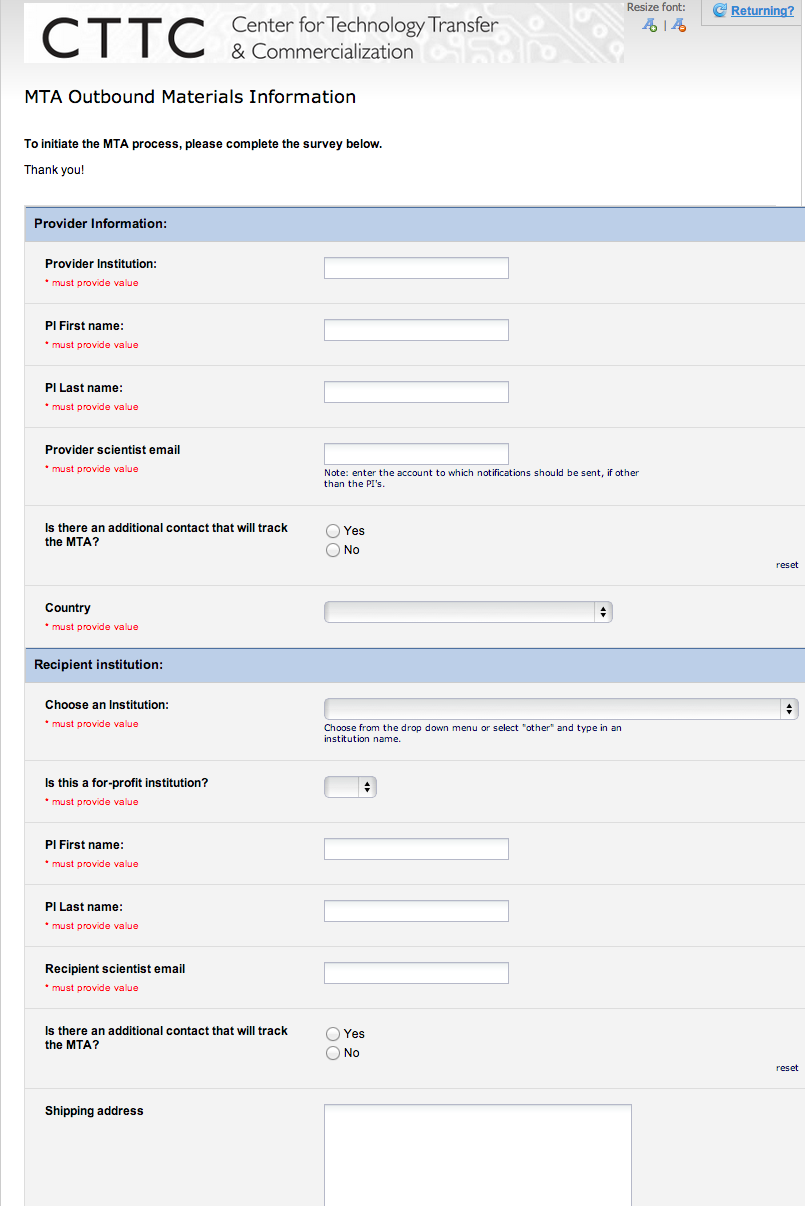
 A month after its official launch, MTAShare has been utilized in the transfer of dozens of research tools developed at Vanderbilt to researchers at other not-for-profit institutions. The system now accommodates the transfer of most any type of material – genetically modified organisms (GMOs), human tissue, stem cells, etc., and utilizes nationally standardized MTAs from the NIH, AUTM, and the UBMTA.
A month after its official launch, MTAShare has been utilized in the transfer of dozens of research tools developed at Vanderbilt to researchers at other not-for-profit institutions. The system now accommodates the transfer of most any type of material – genetically modified organisms (GMOs), human tissue, stem cells, etc., and utilizes nationally standardized MTAs from the NIH, AUTM, and the UBMTA.
It now takes only a few moments for providing scientists to fill out a survey initiating the process, and reviewing draft MTAs administratively takes almost no time at all, except for some of the highly unique or complex MTAs. Many institutions have heard about the benefits of this automation system and have inquired as to how they could gain access to the system. Fortunately, that’s coming.
Currently, we have initiated external testing of MTAShare with about a half dozen not-for-profit institutions. Together, we are testing the transfer of MTAs into and out of our institutions. One challenge we’ve encountered is the variety of internal processes that different institutions employ to review, process and execute MTAs. By testing the system with a diverse user group, we can further contemplate specific features that address most institutional needs and make the system more widely adaptable. We expect this testing phase to end in June, and to make the system broadly available by the 4th of July holiday.
In addition to the external testing, we are also finalizing plans for MTAShare 2.0. This next generation system, scheduled for this fall, will introduce a variety of new features and enhancements, such as a requestor-initiated front end to the system that will save the providing scientist the effort of gathering shipping and use information for material transfers.
Click here to learn more about MTAShare.




